What is Dry Macular Degeneration?
Are you over age 55? Have you noticed a gradual, painless distortion or loss of central vision? Have you noticed color distortion? One possible cause for these visual symptoms is Dry Age-Related Macular Degeneration (ARMD).
Dry Macular Degeneration is the most common type of ARMD, accounting for over 80% of ARMD cases. It leads to a mild-to-moderate loss of sight, although
Peripheral Vision
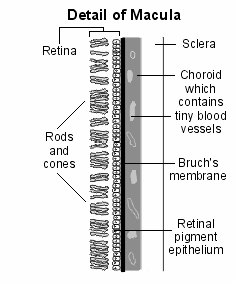
is spared. Dry Macular Degeneration often manifests as light yellow material called drusen located underneath the central retina.
Dry Macular Degeneration is a complex
Eye Disease
affected by multiple external/environmental factors and internal/genetic influences. The exact mechanism for the condition is not fully understood.
In order to view the content, you must install the Adobe Flash Player. Please click
here to get started.
The abnormalities noted in Dry Macular Degeneration appear to begin and progress through Bruch’s Membrane, involving the Retinal Pigment Epithelium (RPE) and photoreceptors. (See diagram above). At the root of the damage seen in Dry ARMD is the aging process with its related oxidative stress.
Oxidative damage occurs from an imbalance between the production and manifestation of highly reactive molecules, known as free radicals, and a biological system’s ability to readily detoxify these free radicals or easily repair the resulting damage.
Free radicals interact with other molecules within cells and cause damage to proteins, membranes and genes in the process. Oxidative damage has been associated with the causes of many diseases such as cancer and Alzheimer’s.
There are external factors that can trigger the production of free radicals such as sunlight and smoking, which also have been implicated in Dry Macular Degeneration.
In the eye, the following can contribute to oxidative stress:
Incoming light, focused on the Macula by the Cornea and Crystalline LensByproducts (or waste products) of cells with high oxygen consumption and energy demandExposure to systemic toxins circulating through the rich blood supply of the ChoroidBoth the breakdown of lipids and proteins in aging photoreceptors and RPE cells aggravates the compromised cellular metabolism and leads to the accumulation of lipofuscin and other waste products, further damaging the cells in charge of their removal.
In turn, this stimulates an immune response, largely mediated by the complement system, further contributing to local damage and beginning a vicious cycle eventually leading either to “quiet” degenerative loss of RPE cells and photoreceptors or Choroidal Neovascularization (CNV) followed by its leaking and bleeding complications, often resulting in severe and sometimes sudden and dramatic visual loss. Thus, Dry ARMD patients are at risk for developing Wet ARMD, which can lead to more damage.
What are signs and symptoms of Dry ARMD?
The earliest clinical signs of Dry ARMD are drusen and macular pigmentary atrophy.
Drusen does not indicate ARMD but is a warning sign that there is the potential for progression and visual loss. ARMD happens to both eyes in over 50% of cases.
The main protein present in drusen is beta-amyloid, which is also present in other chronic age-related neurodegenerative diseases, including Alzheimer’s disease (AD) plaques.In AD, beta-amyloid induces destructive activity of the local immune cells leading to neuronal loss.Dry Macular Degeneration includes the spectrum of findings from minimal drusen through Geographic Atrophy (GA) with no evidence of CNV. See photos below. Some of the other clinical retinal signs include:
Granular clumping and disorganization of the RPE in the Macula areaMacular RPE hyperplasia and degeneration of the outer retinal layers with circumscribed areas of GA of the RPE.Visual symptoms depend on the severity and type of ARMD. In general, Dry Macular Degeneration symptoms are less severe and may include:
Gradual, painless distortion or loss of central visionSome patients complain of color distortion
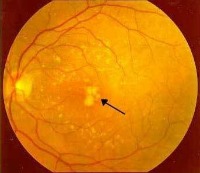
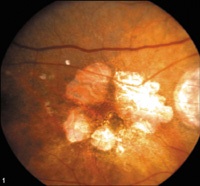
GA is associated with the accumulation of lipofuscin in the RPE. Since the RPE provides nutrients and waste processing for retinal cells, accumulation of lipofuscin in the RPE can lead to death of retinal cells and subsequent loss of vision.
How is Dry ARMD diagnosed?
The best approach in diagnosing and managing Dry Macular Degeneration is to get routine annual or biannual
eye exams
by your eye doctor. Your eye doctor will discuss with you any associated risk factors you may have in experiencing progression and vision loss.
Risk factors include:
Strong association with ageFamily history of ARMDLight complexionHistory of Cardiovascular problemsHistory of previous Lung infection
Hyperopia
Decreased hand grip strengthTypically more progressive in males, but female gender is a risk factorSmoking is a significant risk factorObesity: It has been reported that increased body weight increases oxidative stressMarkers of inflammation are associated with increased BMILow dietary, serum and tissue levels of
lutein and zeaxanthin
Amsler Grid.
If you are diagnosed with early Dry Macular Degeneration, your goal will be to lower your risk of progression:Eliminate potentially harmful ultraviolet light using UV coatings on spectacles and sunglasses. This may also reduce the risk of photochemical/oxidative damage to the retina.Use of antioxidants may play a role in reducing retinal damage by terminating the chemical reactions that are caused by free radicals, created by retinal metabolism.Maintaining a healthy weight with
proper nutrition
and adequate fitness activities.
How is Dry ARMD treated or managed?
Since ARMD is a multifactorial Eye Disease, it has to be attacked by multiple methods. Although the
Experimental Treatments
for ARMD that are available have been studied in small sample sizes and for a short duration, they are all attempting to alter the same common pathway albeit a different step on the pathway.
Areas where research is concentrating are:
Controlling the normal visual cycle such that there is a better balance between waste products produced and the visual system’s removal of them. Fenretinide was shown in pre-clinical studies to reduce the amount of lipofuscin in the eye. It is being investigated as a treatment for GA in Dry Macular Degeneration. Although rods are designed to work during the night, they continue working even during the day. During light exposure, rods generate by-products such as lipofuscin as well as A2E, which is a major component of lipofuscin. Fenretinide has been found to slow the production and accumulation of A2E, a by-product of Vitamin A.ACU-4429 controls the visual cycle by significantly reducing the accumulation of the retina related toxic by-product A2E, believed to damage retinal cells.Blocking of the complement system since this system is possibly involved at all stages of ARMD, the complement inhibitors being developed can potentially work for patients with early as well as advanced ARMD. Some of the complement inhibitors being investigated:
POT-4 is designed to inhibit complement component C3 from participating in the complement activation cascade. C3 can lead to local inflammation, tissue damage and increased receptor numbers for angiogenic factors such as Vascular Endothelial Growth Factor (VEGF). POT-4 is not intended to replace
Lucentis®
but may be used on those who don’t respond to Lucentis® or before the need for Lucentis® arises. Eculizumab is a humanized monoclonal antibody to C5. It is being studied for its efficacy in reducing both the growth rate of GA as well as high-risk drusen area and volume.ARC 1905 is another agent targeting C5. It is being investigated for the treatment of Dry Macular Degeneration and CNV (in combination with an anti-VEGF agent)JPE-1375 is a small molecule that targets the receptor for C5a on inflammatory cells. It is a biodegradable injectable designed to last for 6 months. TNX-234 is a humanized antibody that is directed against a component of the complement system. It is being evaluated for safety in an intravitreal injection for patients with GA. Protecting photoreceptors and neuronal tissueA New York Eye & Ear Infirmary project is investigating in eyes with Dry ARMD the efficacy and safety as preventative therapy of Copaxone, a T cell-based vaccination.Copaxone has been shown to alter microglia (which acts as the first and main form of active immune defense in the brain) into a neuroprotective form which results in reduction of cognitive decline, elimination of plaque formation, and induction of neuronal survival.Copaxone is being studied to find out if its effect on drusen in Dry ARMD is similar to that on deposits of other age-related chronic neurodegenerative diseases such as Alzheimer’s disease.Various delivery methods are being tested, including a weekly vaccination.NT-501 Intraocular Implant delivers human cells that have been genetically altered to release Ciliary Neurotrophic Factor (CNTF).CNTF has been demonstrated to be able to rescue dying photoreceptors in several patterns of neurodegeneration.OT-551 is an experimental drug in
eye drop
form for treatment of Geographic Atrophy by reducing oxidative stress and disease-induced inflammation. It appears to have a positive additive effect when combined with either
Lucentis or Avastin.
It also may have a neuroprotective effect.Introducing
nutrients that benefit the eye
Age-Related Eye Disease Study (AREDS) showed that high levels of antioxidants and zinc significantly reduce the risk of advanced ARMD and its associated vision loss. AREDS2 is a follow-up study evaluating the effects of Oral supplementation of the following agents on the progression to advanced ARMD:
macular xanthophylls (lutein and zeaxanthin) and/or long-chain omega-3 fatty acids:doxosahexaenoic acid-DHA andeicosapentaenoic acid-EPAEliminating beta-carotene and reducing zinc in the original AREDS formulation.Attacking Dry ARMD on four fronts (i.e., controlling visual cycle, complement inhibition, neuroprotection, and nutritional supplementation) may open the pathway to arresting and perhaps even reversing the vision loss caused by this multifactorial Eye Disease.
Return From Dry Macular Degeneration to Macular Degeneration
Enjoy this page? Please pay it forward. Here's how...
Would you prefer to share this page with others by linking to it?
- Click on the HTML link code below.
- Copy and paste it, adding a note of your own, into your blog, a Web page, forums, a blog comment,
your Facebook account, or anywhere that someone would find this page valuable.







